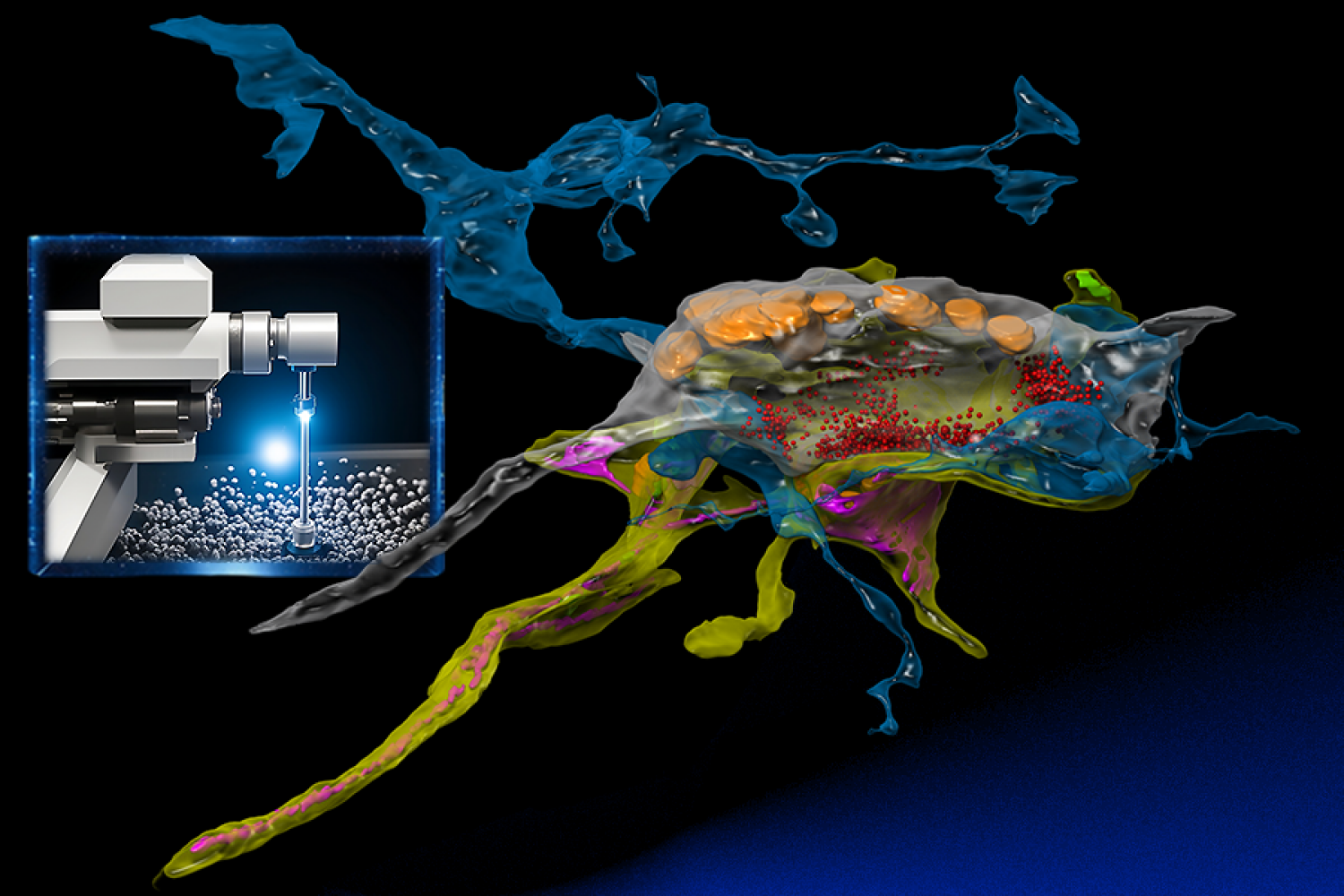
Connectomics, the ambitious field of study that seeks to map the intricate network of animal brains, is well underway. Within a decade, it has traveled from emerging stages to a discipline poised (hopefully) to unlock the mysteries of cognition and the physical underpinnings of neuropathologies such as Alzheimer’s disease.
At its forefront is the use of powerful electron microscopes, which researchers from the MIT Computer Science and Artificial Intelligence Laboratory (CSAIL) and Harvard University’s Samuel and Lichtman Labs have endowed with the analytical power of machine learning. Unlike traditional electron microscopy, the built-in AI serves as a “brain” that learns a sample as the images are captured and intelligently focuses on relevant pixels at nanoscale resolution similar to how animals survey their worlds.
“SmartEMIt helps the synecologist to quickly examine and reconstruct the brain’s complex network of synapses and neurons with nanometer precision. Unlike traditional electron microscopy, its built-in AI opens new doors to understand the complex architecture of the brain.
Hardware and software integration in the process is crucial. The team integrated a GPU into the support computer attached to their microscope. This enabled machine learning models to be run on the images, helping to direct the microscope beam to areas deemed interesting by the AI. “This allows the microscope to linger longer in areas that are harder to understand until it captures what it needs,” says MIT professor and CSAIL principal investigator Nir Shavit. “This step helps mirror the control of the human eye, enabling rapid understanding of images.”
“When we look at a human face, our eyes quickly navigate to focal points that provide vital cues for effective communication and understanding,” says SmartEM lead architect Yaron Meirovitch, a visiting scientist at MIT CSAIL who is also a former postdoc. and current research associate neuroscientist at Harvard. “When we immerse ourselves in a book, we do not scan all the empty space. Instead, we direct our gaze toward words and characters with ambiguity relative to our sentence expectations. This phenomenon within the human visual system paved the way for the birth of the new idea of the microscope.”
For the task of reconstructing a section of a human brain of about 100,000 neurons, achieving this with a conventional microscope would require a decade of continuous imaging and a prohibitive budget. But with SmartEM, by investing in four of these innovative microscopes at less than $1 million each, the work could be completed in just three months.
Nobel and worms
Over a century ago, Spanish neuroscientist Santiago Ramón y Cajal was heralded as the first to characterize the structure of the nervous system. Using the rudimentary light microscopes of his day, he launched groundbreaking explorations in neuroscience, establishing a fundamental understanding of neurons and sketching the initial contours of this vast and uncharted realm—a feat that won him a Nobel Prize. He noted, on the subjects of inspiration and discovery, that “As long as our brain is a mystery, the universe, the reflection of the brain’s structure, will also be a mystery.”
Moving on from these early stages, the field has advanced dramatically, as evidenced by efforts in the 1980s mapping the relatively simpler connection of C. elegans, tiny worms, in current efforts to probe the more complex brains of organisms such as zebrafish and mice. This development reflects not only enormous strides, but also escalating complexities and demands: mapping the mouse brain alone means managing a staggering one thousand petabytes dataa task that greatly overshadows any university’s storage capabilities, the team says.
Water tests
For their own work, Meirovitch and others on the research team studied 30-nanometer-thick slices of octopus tissue that were taped, placed on wafers, and finally inserted into electron microscopes. Each part of an octopus brain, made up of billions of pixels, was imaged, allowing scientists to reconstruct the slices into a three-dimensional cube at nanometer resolution. This provided an extremely detailed view of the synapses. The main goal? To color these images, identify each neuron and understand their interactions, thereby creating a detailed map, or “wireframe,” of the brain’s circuitry.
“SmartEM will reduce the imaging time of such projects from two weeks to 1.5 days,” says Meirovitch. “Neuroscience laboratories that are currently unable to engage in precise and long-term EM imaging will now be able to do so,” The method should also enable circuit analysis at the synaptic level in samples from patients with psychiatric and neurological disorders.
The team then envisions a future where connectomics is both affordable and accessible. They hope that with tools like SmartEM, a wider range of research institutions could contribute to neuroscience without relying on large collaborations, and that the method will soon be standard line in cases where biopsies from living patients are available. Additionally, they are eager to apply the technology to understand pathologies, extending the utility beyond simple linker genomics. “We are now trying to introduce it into hospitals for large biopsies, using electron microscopes, with the aim of making pathology studies more efficient,” says Shavit.
Two other authors on the paper have ties to MIT CSAIL: lead author Lu Mi MCS ’19, PhD ’22, who is now a postdoctoral fellow at the Allen Institute for Brain Science, and Shashata Sawmya, an MIT graduate student in the lab. The other lead authors are Core Francisco Park and Pavel Potocek, while Harvard professors Jeff Lichtman and Aravi Samuel are additional senior authors. Their research was supported by the NIH BRAIN Initiative and was presented at the International Conference on Machine Learning (ICML) 2023 for Computational Biology. The work was done in collaboration with scientists from Thermo Fisher Scientific.